Cancer drug restores immune system’s ability to fight tumors
Researchers have pioneered a novel, bio-inspired drug that reinvigorates the immune system’s capability to fight various cancers.
[Oct. 22, 2023: Staff Writer, The Brighter Side of News]
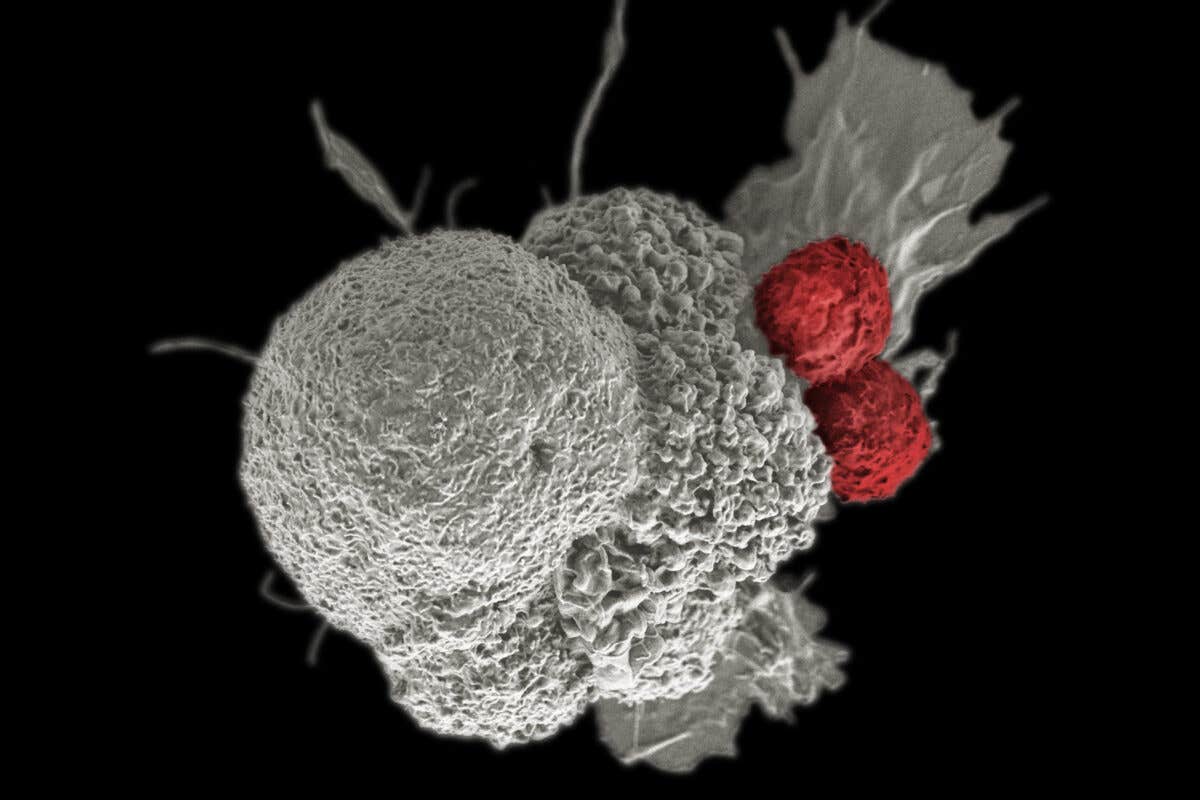
A pseudo-colored scanning electron micrograph of an oral squamous cancer cell (white) being attacked by two cytotoxic T cells (red). (CREDIT: Rita Elena Serda, Duncan Comprehensive Cancer Center at Baylor College of Medicine, National Cancer Institute, National Institutes of Health)
In a groundbreaking discovery, scientists from The University of Texas at Austin have pioneered a novel, bio-inspired drug that reinvigorates the immune system's capability to fight various cancers.
This innovation, tested in mouse models of melanoma, bladder cancer, leukemia, and colon cancer, not only decelerates tumor growth and prolongs life but also amplifies the effectiveness of existing immunotherapy treatments.
Published in the esteemed journal Cancer Cell, this research is poised to radically alter the therapeutic landscape for many cancer patients, potentially shifting the paradigms of cancer treatment.
"Cancers frequently exhibit a deletion in a segment of DNA known as 9p21, the most prevalent deletion spanning all cancer types," explains Everett Stone, research associate professor in the Department of Molecular Biosciences and associate professor of oncology at Dell Medical School, who spearheaded the research initiative.
Related Stories
This genetic anomaly is present in 25% to 50% of certain cancer types, including melanoma, bladder cancer, mesothelioma, and particular brain cancers.
For years, the scientific community has recognized that cancers harboring the 9p21 deletion are synonymous with poorer patient outcomes and pronounced resistance to immunotherapies. These therapies, conceptualized to enhance a patient’s innate immune response to cancer, often falter against these genetically cunning cancer cells.
The insidiousness of the 9p21 deletion lies in its enabling the cancer cells to evade detection and destruction by the immune system. This is achieved by triggering the cancer cells to release a noxious substance, Methylthioadenosine (MTA), which not only debilitates the normal functions of immune cells but also diminishes the potency of immunotherapies.
MTAP loss increases MTA levels and ICI failure in melanoma and urothelial tumors. (CREDIT: Cancer Cell)
Stone elaborates on the drug's mechanism: “In animal models, our drug mitigates the levels of MTA, restoring them to normalcy, and consequently, the immune system is reactivated. We observe a significant increase in T cells surrounding the tumor, and they are primed for attack. T cells are crucial immune sentinels, akin to a SWAT team, proficient in identifying tumor cells and annihilating them from within."
Envisioning the future of this drug, Stone anticipates it will be utilized in tandem with immunotherapies to heighten their impact. The profound implications of this research are not lost on the cancer community, as it opens avenues for treatments that could be more effective and tailored to the genetic makeup of the cancer.
PEG-MTAP delivery to MTAP−/− tumors controls tumor growth dependent of CD8+ T cells. (CREDIT: Cancer Cell)
The intricacies of the 9p21 deletion are such that it results in the eradication of vital genes within cancer cells. Particularly, it eliminates a duo of genes responsible for the production of cell cycle regulators — proteins integral to maintaining the measured growth and division of healthy cells. In their absence, cellular growth becomes unbridled, a hallmark of cancerous transformation. Concurrently, the deletion expunges a "housekeeping" gene tasked with encoding an enzyme that neutralizes the toxin MTA.
"Cancer obtains a dual advantage with this single genetic loss," Stone remarks. "It effectively removes the inherent restrictions that prevent rampant growth, while simultaneously disabling the body’s internal defense mechanisms. This results in a more virulent and aggressive cancer phenotype."
To engineer their drug candidate, Stone and his team initiated their process with the naturally occurring enzyme responsible for neutralizing MTA, augmenting it with flexible polymers for stability and longevity.
Stone explains the optimization process: "The enzyme in its natural form is highly efficient, but we needed to enhance its durability within the body. The unaltered enzyme, if injected, would dissipate within a few hours. However, our modified variant persists in the mice's circulation for days, and we predict an even longer circulation time in humans."
Named PEG-MTAP, the drug will undergo additional safety evaluations, with the research team actively pursuing funding for human clinical trials, marking a significant step forward in the translation of this research from bench to bedside.
The study’s collaboration involved co-first authors Donjeta Gjuka, a former UT postdoctoral researcher now at Takeda Oncology, and Elio Adib, previously with Brigham and Women’s Hospital and the Dana-Farber Cancer Institute and currently a resident physician at Mass General Brigham. Other notable contributors from UT include Kendra Garrison, Candice Lamb, Yuri Tanno, and George Georgiou. Co-corresponding authors alongside Stone were David Kwiatkowski, a senior physician at Brigham and Women's Hospital and a professor of medicine at Harvard Medical School.
Excess MTA in the TME impairs CD4+ and CD8+ T cells via ADO agonism and PRMT5 inhibition. (CREDIT: Cancer Cell)
This monumental work received financial support from several institutions, including the National Cancer Institute, the Doris Duke Foundation, The University of Texas MD Anderson Cancer Center, the Joan and Herb Kelleher Charitable Foundation, the Kidney Cancer Association, the V Foundation, and the U.S. Department of Defense.
The study underscores the potential of bio-inspired drugs in overcoming genetic hurdles in cancer treatment, providing new hope for patients and adding a critical weapon to the arsenal in the war against cancer. The perseverance and ingenuity of these researchers underscore the relentless human endeavor to understand and conquer this complex disease. Their findings are not just a scientific triumph, but a beacon of hope for patients worldwide.
Note: Materials provided above by The Brighter Side of News. Content may be edited for style and length.
Like these kind of feel good stories? Get the Brighter Side of News' newsletter.