Lifechanging technology could reduce daily diabetes injections to just three shots a year
Scientists have unveiled a pioneering hydrogel drug delivery system capable of transforming diabetes and weight control drug delivery
[Dec. 1, 2023: JD Shavit, The Brighter Side of News]
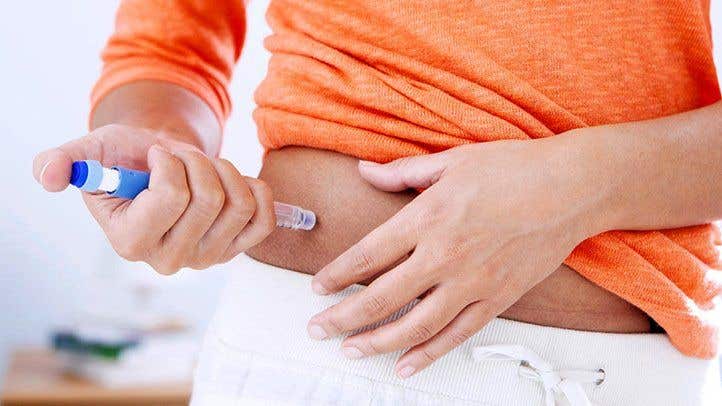
Scientists have unveiled a pioneering hydrogel drug delivery system capable of transforming the way diabetes and weight control drugs are administered. (CREDIT: Creative Commons)
In a groundbreaking development, materials engineers at Stanford University have unveiled a pioneering hydrogel drug delivery system capable of transforming the way diabetes and weight control drugs are administered.
The innovative system, detailed in a recent study published in Cell Reports Medicine, holds the potential to significantly enhance the management of both diabetes and obesity, promote better patient compliance, and improve long-term health outcomes for individuals with Type 2 diabetes.
Currently, medications like Ozempic, Mounjaro, Trulicity, Victoza, and others that help regulate blood sugar levels and control weight operate by mimicking the hormone glucagon-like peptide 1 (GLP-1). While these drugs have proven highly effective in aiding individuals in managing their diets and weight, the need for daily or weekly injections presents a substantial burden for many patients.
Eric Appel, an associate professor of materials science and engineering at Stanford and the principal investigator behind the groundbreaking hydrogel, emphasized the challenge of adherence in Type 2 diabetes management. He remarked, “Needing only three shots a year would make it much easier for people with diabetes or obesity to stick with their drug regimens.”
Related Stories
Type 2 diabetes is a global health concern, affecting half a billion people worldwide, with a staggering 130 million individuals grappling with the condition in the United States alone. The economic toll of treating Type 2 diabetes is estimated to exceed $400 billion annually in the United States.
GLP-1 drugs, hailed as "miracle drugs" due to their minimal side effects and profound impact on energy intake regulation, have garnered attention for their ability to help patients feel more satiated, less hungry, and address other reward-related dietary effects.
The Innovative Nanocomposite Hydrogel
At the heart of this revolutionary drug delivery system lies a unique hydrogel with distinct physical properties. While hydrogels themselves are not new, Appel's hydrogel distinguishes itself by utilizing polymers and nanoparticles that are loosely bound to one another, allowing it to function as a gel while gradually releasing the encapsulated drugs over an extended period. The hydrogel is constructed from a mesh of polymer chains and nanoparticles that securely retain the drug molecules until the mesh slowly dissolves, releasing the drugs into the body.
Graphical abstract: Hydrogel depot technology enables a months-long GLP-1 drug product. (CREDIT: Cell Reports Medicine)
Appel explained, "Our hydrogel melts away over many months like a sugar cube dissolving in water, molecule by molecule. I often refer to the mesh being held together by a sort of molecular Velcro that sticks together quite well but can then be easily pulled apart."
This innovative hydrogel, technically referred to as a polymer-nanoparticle (PNP) hydrogel, boasts the ideal balance of fluid-like flow that enables straightforward injection using off-the-shelf needles while maintaining a gel-like stability durable enough to last the full four-month release period. GLP-1 drug molecules are seamlessly integrated into the hydrogel, which then releases them gradually as it slowly dissolves.
PNP hydrogels for the prolonged delivery of GLP-1 RAs. From literature reports it is clear that once-weekly dosing frequency does not significantly improve patient compliance compared with a once-daily dosing frequency. (CREDIT: Cell Reports Medicine)
The administration of the hydrogel is relatively simple, involving the injection of a small, drug-laden gel depot under the skin, typically in a convenient location such as under the arm. The key challenge for the engineers was to design the hydrogel in a manner that ensures the depot remains small enough to be comfortable and inconspicuous for the patient while being large and robust enough to provide the intended four-month duration of drug release. Appel expressed confidence that his team has successfully achieved this balance.
"We chose four months to match the cadence that people actually meet with their physician or endocrinologist, and why we were so specific with the release period," Appel remarked.
Initial tests of the new drug delivery system in laboratory rats have yielded highly promising results. In these trials, a single injection of the hydrogel-based therapy demonstrated superior efficacy in managing blood glucose levels and weight when compared to daily injections of a leading commercial drug, as highlighted by Appel.
Preparation and characterization of GLP-1 RA-loaded PNP hydrogel formulations. PNP hydrogels are prepared by mixing a solution of hydrophobically modified HPMC (right syringe) with a solution of PEG-PLA nanoparticles and GLP-1 RAs (left syringe) using a Luer lock mixer. (CREDIT: Cell Reports Medicine)
While this hydrogel was originally tailored for the four-month GLP-1 regimen, Appel noted that the team has successfully adjusted the release timeframes to range from just days to an impressive six months. He further pointed out that similar systems have been utilized for delivering other proteins, vaccines, and therapeutic cells, with evidence suggesting that GLP-1 drugs may also reduce the risk of cardiovascular disease. These promising indications strongly suggest that this drug delivery system may be adaptable to a wide array of medications and conditions.
Appel spoke optimistically about the potential applications of this system, stating, "There's even been really promising results with children with Type 1 diabetes," underscoring the bright prospects ahead.
In vitro evaluation of PNP hydrogel formulation release kinetics. Schematic of the in vitro release assay of GLP-1 RAs from PNP hydrogels immersed in saline over 2 weeks. (CREDIT: Cell Reports Medicine)
The next step in the development of this innovative technology involves testing it on pigs, whose skin and endocrine systems closely resemble those of humans. If these trials proceed as planned, human clinical trials could be on the horizon within a year and a half to two years.
"At the very least, we have laid a pathway for the prolonged release of therapeutic GLP-1–based anti-diabetic and anti-obesity treatments that could have a beneficial impact on Type 2 diabetes management and, perhaps, other conditions as well," Appel affirmed.
As further research and clinical trials progress, this innovative technology may hold the key to more effective drug delivery for a range of medical conditions, promising a brighter future for patients worldwide.
Note: Materials provided by Université de Genève. Content may be edited for style and length.
Like these kind of feel good stories? Get the Brighter Side of News' newsletter.



