New hydrogel prints bone-like implants at record speed
ETH Zurich researchers printed a jelly-like hydrogel scaffold that mimics bone’s channels and may support future implants.

 Edited By: Joseph Shavit
Edited By: Joseph Shavit
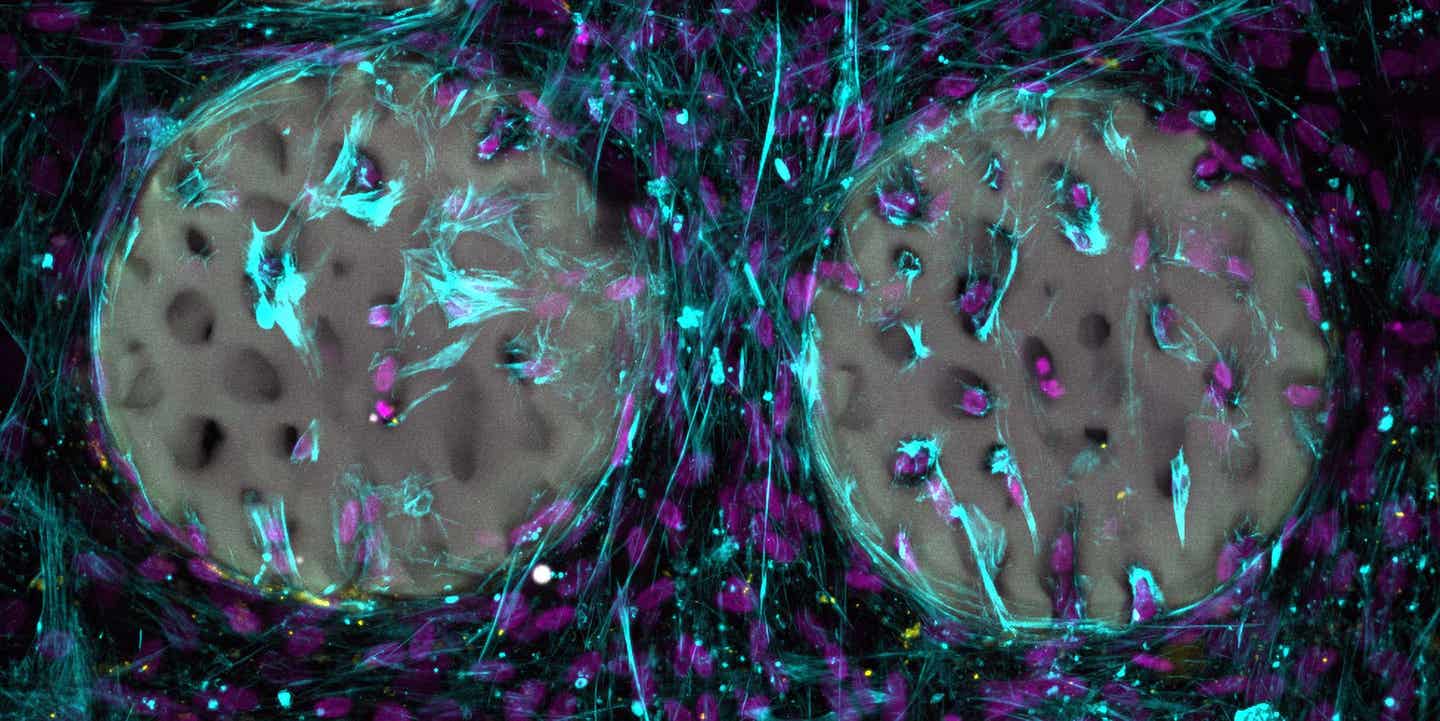
A good sign: Bone-forming cells (light blue) with nuclei (purple) have already colonized a hydrogel that has been structured like bone. (CREDIT: Margherita Bernero / ETH Zurich)
A cube of healthy bone is anything but solid. Inside it, countless tiny channels carry fluid and help cells move, feed, and rebuild. Xiao-Hua Qin, a professor of biomaterials engineering at ETH Zurich, likes a striking comparison: “A piece of bone the size of a dice contains 74 kilometers of tunnels.” The Gotthard Base Tunnel, the world’s longest railway tunnel, runs 54 kilometers.
That hidden architecture is one reason broken bones usually heal, and one reason severe breaks can become a surgical puzzle.
When a fracture is too extensive, or when surgeons remove a bone tumor, they often rely on implants that help the body knit the gap back together. Many of today’s options involve autografts, pieces of a patient’s own bone, or metal and ceramic parts. Autografts can require a second surgery to harvest tissue. Metal implants, meanwhile, can be too rigid and may loosen over time, which can compromise stability.
Qin and colleagues are now pushing a different idea: an implant material that starts soft, like the body’s own first step in repair, and can be printed into intricate bone-like shapes.
Bone repair starts squishy
Early bone healing does not begin with something hard. In the first days after a fracture, the body forms a haematoma, essentially a bruise, that is permeable. It allows reparative and immune cells to migrate, and it helps deliver nutrients. A fibrin network holds the cells together. Only later does that soft structure transform into hard, stiff bone.
That sequence shaped the strategy behind a new hydrogel developed at ETH Zurich by Qin, his team, and ETH Professor Ralph Müller. The hydrogel is as soft as jelly and dissolves gradually in the body. The researchers describe it as a potential material for personalized bone implants, and they report their work in the journal Advanced Materials.
“For proper healing, it is vital that biology is incorporated into the repair process,” Qin said. A successful repair depends on various cell types that first need to colonize an implant before forming new bone tissue.
Hydrogels have long appealed to tissue engineers because they can mimic soft biological environments. They also frustrate engineers for the same reason. Soft gels are hard to shape into stable, detailed structures.
Printing jelly at extreme speed
The ETH hydrogel is mostly water, 97 percent, with 3 percent biocompatible polymer. To make it solidify in precisely chosen spots, the researchers introduced two molecules. One links polymer chains together. The other triggers the linking reaction when exposed to light.
Wanwan Qiu, a former doctoral student of Qin and Müller, developed the connecting molecule for this application. “It enables rapid structuring of hydrogels in the sub-micrometer range,” she said.
Here is how the shaping works in practice. Laser pulses of a certain wavelength hit the hydrogel and link the polymer chains in the targeted region. Those irradiated areas become solid right away. Non-irradiated material can be washed out later.
That approach lets the team “print” structures into the gel with fine resolution and high precision. The smallest structures they report are about 500 nanometers.
“Hydrogels resemble jelly, making them difficult to shape,” Qin said. “With our newly developed connecting molecule, we can now not only structure the hydrogel in a stable and extremely fine manner but also produce it at high writing speeds of up to 400 millimeters per second. That’s a new world record.”
The method behind the printing is called two-photon polymerization, or 2PP, a direct laser writing technique used to fabricate high-resolution 3D hydrogel architectures for tissue engineering and drug delivery. It uses very short laser pulses to trigger solidification in a tiny focal volume, giving strong spatial control compared with single-photon photolithography.
Why chemistry mattered this time
The technical challenge was not just getting small features. The researchers also wanted soft, extracellular-matrix-like gels that still print cleanly.
In 2PP, many strategies rely on high polymer concentrations, often 20 to 50 percent, to achieve printability at low writing speeds. Yet low polymer concentration helps mimic the low stiffness of native tissues.
The ETH team focused on a problem that can show up in thiol-ene hydrogels made with common small crosslinkers. At low polymer concentrations, a crosslinker can react in ways that create “primary loops,” a kind of defect caused by intramolecular crosslinking. Those defects reduce network connectivity and can compromise gel stability.
Their solution was to design a macromolecular thiol crosslinker called PVASH, meant to improve crosslinking efficiency and reduce those loop defects. They paired it with norbornene-functionalized polyvinyl alcohol, or nPVA, and used specific photoinitiators depending on whether they used one-photon or two-photon excitation.
Mechanical and swelling tests
In mechanical and swelling tests, PVASH-based hydrogels formed stable gels at lower concentrations than some reference crosslinkers. The team reports storage moduli spanning from 0.1 to 10 kPa, depending on concentration, and they measured faster gelation onset times for PVASH than for certain other crosslinkers at low precursor concentrations.
They also tested how PVASH hydrogels held up in buffer. Degradation depended strongly on pH. At pH 10, the compressive modulus dropped sharply over days, the swelling ratio increased dramatically, shape fidelity was lost, and the gels completely degraded by day 14. At pH 7.4, the modulus declined more modestly with negligible swelling changes.
On the printing side, the group mapped a “processing window” by varying laser power and scanning speed. For a 4 percent formulation, PVASH showed a larger processing window than a PEG-based reference, and the researchers printed several complex 3D structures, including a femur model and gyroid patterns. They also note that at 3 percent polymer concentration, PVASH remained printable in a large window while the PEG-based formulation became poorly printable.
At 2 percent polymer content, printability declined because crosslinking density was insufficient. The team tested an idea borrowed from a “double-network” concept by adding 3 percent gelatin as a sacrificial network, which improved printability of the 2 percent PVASH formulation. They report the printed structures stayed stable after two days at 37°C, and they note that more than 80 percent of gelatin can diffuse out based on a previous report.
Cells liked the scaffolds, with caveats
So far, the work stays in the lab. The ETH researchers tested the material in a test tube.
They evaluated PVASH macromer solutions using an MTS cell proliferation assay with human dermal fibroblasts. Cell viability remained high, above 95 percent, after 24 hours of incubation.
To help cells stick to the printed micro-scaffolds, the team used RGD-functionalized nPVA conjugates. Control experiments showed that cells did not adhere to micro-scaffolds lacking RGD, which underscored the need for those motifs.
On woodpile micro-scaffolds with 10 micrometer pores, fibroblasts spread extensively, and some formed networks through long protrusions. In contrast, far fewer cells were found on 25 micrometer pore scaffolds, which the researchers suggest may be because the pores were too large to retain cells from sedimentation.
The team also cultured human mesenchymal stromal cells on a 10 micrometer woodpile scaffold and on trabecular femur constructs under osteogenic conditions for up to 14 days. They observed cells interacting with the micro-scaffolds, contracting, clustering, and self-assembling into dense structures around the scaffolds. The researchers flag a limitation here: heterogeneous cell densities across samples, which may complicate functional readouts. They suggest that incorporating a supporting 3D matrix could improve future cultures by promoting more uniform cell distribution.
The paper also lists unanswered questions. The exact conversion degree of thiol groups after 2PP still needs testing, to compare defects across different hydrogel architectures. The team also calls for more work on mechanobiological principles behind cell-scaffold interactions, biodegradation, and long-term biocompatibility.
Qin’s declared goal is clinical use for bone repair, but the next steps come first. He is preparing animal tests with the AO Research Institute Davos, to see whether the material promotes migration of bone-forming cells in living organisms and whether it restores bone strength over time.
Practical implications of the research
If this approach holds up beyond the test tube, it could shift how some bone implants get designed.
A material that starts soft, prints into bone-like micro-architecture, and gradually dissolves could offer surgeons a scaffold that invites cells in early, rather than forcing them to adapt to a rigid surface.
The printing speed and fine feature control could also make it easier to tailor implants to a patient’s imaging data, especially for complex defects where shape and internal channels matter as much as outer contours.
Research findings are available online in the journal Advanced Materials.
The original story "New hydrogel prints bone-like implants at record speed" is published in The Brighter Side of News.
Related Stories
- Scientists use light-controlled nanorobots to quickly grow bone cells
- 3D printing 'glue gun' heals fractures by printing living bone in real time
- Scientists grow real teeth in the lab - replacing fillings and implants
Like these kind of feel good stories? Get The Brighter Side of News' newsletter.
Mac Oliveau
Writer
Mac Oliveau is a Los Angeles–based science and technology journalist for The Brighter Side of News, an online publication focused on uplifting, transformative stories from around the globe. Passionate about spotlighting groundbreaking discoveries and innovations, Mac covers a broad spectrum of topics including medical breakthroughs, health and green tech. With a talent for making complex science clear and compelling, they connect readers to the advancements shaping a brighter, more hopeful future.



