Revolutionary CAR T cell therapy represents new hope for millions of cancer patients
[Sept. 21, 2023: Staff Writer, The Brighter Side of News] Chimeric antigen receptor T cell (CAR T) therapy has delivered promising…
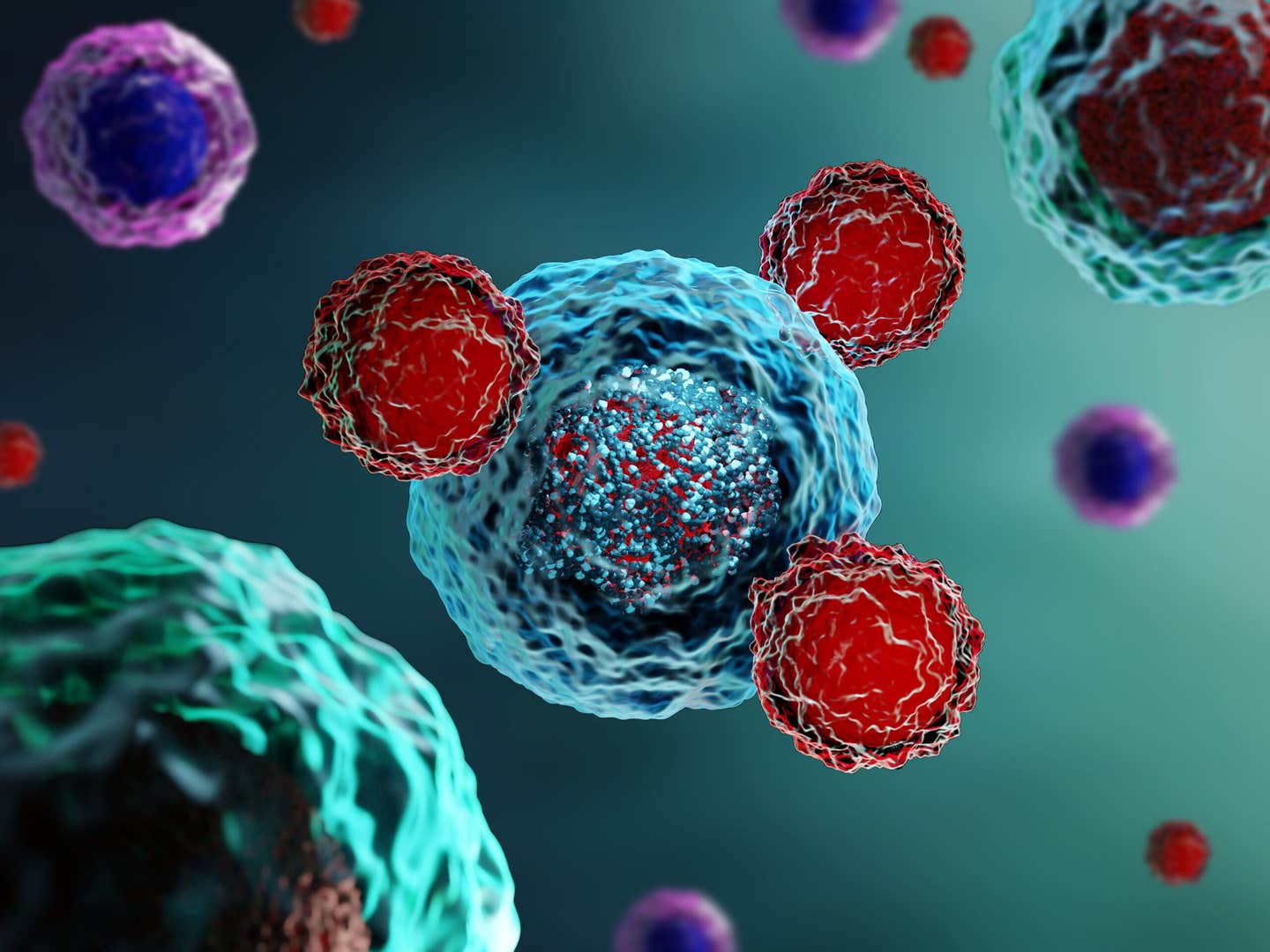
[Sept. 21, 2023: Staff Writer, The Brighter Side of News]
Chimeric antigen receptor T cell (CAR T) therapy has delivered promising results, transforming the fight against various forms of cancer, but for many, the therapy comes with severe and potentially lethal side effects. Now, a research team led by Michael Mitchell of the School of Engineering and Applied Science has found a solution that could help CAR T therapies reach their full potential while minimizing severe side effects. (CREDIT iStock / Meletios Verras)
In the sprawling landscape of oncological research, one groundbreaking therapy, the chimeric antigen receptor T cell (CAR T) therapy, stands tall. With its potential to transform the treatment trajectory of various cancers, it represents the next frontier of hope for millions.
However, like any pioneering solution, it comes with challenges – some as severe as life-threatening side effects. Now, a fresh approach led by a team from the University of Pennsylvania promises to not only counter these side effects but potentially open new horizons for cellular therapies.
A Promising Dawn: The Rise of CAR T
For those unfamiliar with the nuances of cancer therapy, CAR T therapy has emerged as nothing short of revolutionary. This procedure modifies a patient's own T-cells, making them capable of targeting and destroying cancer cells. The result? Success rates that are astonishingly high for certain types of cancers which were once deemed almost untreatable.
PEGylation of CAR T cells alters cell-to-cell interactions and cytokine release. (CREDIT: Nature Materials)
The US Food and Drug Administration (FDA) has already greenlit six CAR T cell therapies, and the clinical pipeline is brimming with others eagerly awaiting approval. However, as is often the case with cutting-edge medical breakthroughs, there's a flip side.
Cytokine release syndrome (CRS) and neurotoxicity are two severe side effects accompanying CAR T therapy. Patients might experience a range of symptoms from these effects, spanning from high fever and vomiting to more severe ones like multiple organ failure. In some tragic instances, death is the consequence. This obviously raises pertinent concerns about the broader clinical application of CAR T therapies.
In the words of Michael Mitchell, an associate professor at the University of Pennsylvania's School of Engineering and Applied Science and the lead researcher on the latest study, “Addressing CRS and neurotoxicity without compromising the therapeutic effectiveness of CAR T cells has been a complex challenge.”
Related Stories
Mitchell further elaborates that the complications arise from the unwanted interactions between the CAR T cells and a type of immune cell known as macrophages. These interactions induce the macrophages to release toxic cytokines, culminating in CRS and neurotoxicity.
But where biology poses challenges, engineering provides solutions. And Mitchell's team has done precisely that.
Describing the approach, Mitchell said, “Our study introduces a materials engineering-based strategy. We incorporate a sugar molecule onto the CAR T cells' surface.” This modification allows the cells to retain their ability to fight cancer. However, the magic happens when these sugars act as reactive handles, enabling the creation of a protective biomaterial coating or what Mitchell metaphorically describes as a ‘suit of armor’. This armor safeguards the CAR T cells from dangerous macrophage interactions.
Mechanistic illustration of the in situ PEGylation strategy for managing cytokine release syndrome and neurotoxicity. (CREDIT: Nature Materials)
Ningqiang Gong, the study's first author and a postdoctoral researcher in the Mitchell Lab, detailed the process. “When symptoms of CRS begin to manifest, we introduce another molecule—polyethylene glycol (PEG)—to create the suit of armor. This blocks perilous interactions among the engineered T cells, macrophages, and even tumor cells.”
But this isn’t a permanent barrier. Over time, smaller tumor antigens can interact with these ‘PEGylated CAR T cells’. This slow engagement avoids the catastrophic side effects linked with rapid activation. As these CAR T cells gradually expand, the PEG on their surface dilutes, gradually restoring their interaction capabilities.
In situ PEGylation of CAR T cells abolishes neurotoxicity. (CREDIT: Nature Materials)
The beauty of this solution lies in its nuanced approach to treating cancer. Gong explains that due to size differences between tumor cells, CAR T cells, and macrophages, interactions between CAR T cells and tumor cells are restored before those with macrophages. This means CAR T cells can annihilate cancer cells without triggering dangerous CRS symptoms and neurotoxic effects.
This protective PEG buffer thus achieves a delicate balance. “By incorporating the PEG buffer, we’ve successfully managed to modulate the interactions between CAR T cells and macrophages. This enables a therapy that is both safer and effective,” Mitchell remarked.
Conjugation of DBCO-PEG600K to CAR T cells in vivo. (CREDIT: Nature Materials)
The implications of this study extend far beyond just CAR T therapy. The team envisions applying in situ PEGylation to other cellular immunotherapies and possibly even more extensive medical applications. Mitchell concludes with a note of optimism, stating, “We’re looking at a potentially universal approach to making cellular therapies safer for all patients.”
As this research progresses, it's clear that the intersection of biology and engineering might hold the key to making the promises of CAR T therapy a safer reality for cancer patients worldwide. Only time will tell how far this 'suit of armor' can take the medical community in its relentless fight against cancer.
Note: Materials provided above by The Brighter Side of News. Content may be edited for style and length.
Like these kind of feel good stories? Get the Brighter Side of News' newsletter.