Hard truth: Here’s the reason most lithium-ion batteries fail
A new study overturns decades of assumptions about lithium dendrites, finding they are brittle and rigid, with major implications for battery safety and design.

 Edited By: Joseph Shavit
Edited By: Joseph Shavit
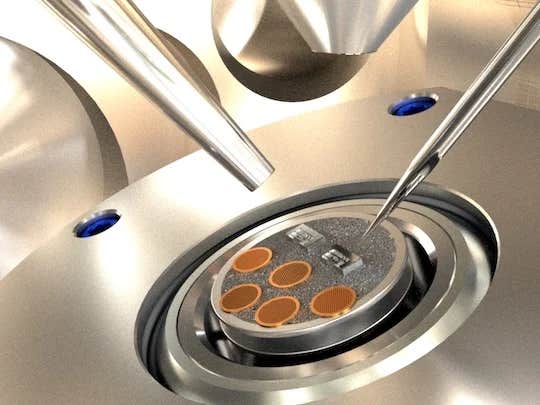
Close-up view of the top of the sample transfer box (top door open), showing that the lithium dendrite was transferred using a micromanipulator tip (a sharp silver needle) from the brown copper transmission electron microscopy grids to the Rice micromechanical devices (silver blocks), ready for subsequent testing and characterization. (CREDIT: Lou Group/Rice University)
Lithium was supposed to be soft. The metal bends easily in bulk form, stretches before it breaks, and deforms the way you might expect a pliable material to behave. Scientists who studied the tiny thorn-like structures that grow inside lithium-ion batteries assumed those structures would behave the same way. They were wrong.
A new study published in the journal Science has found that lithium dendrites, the microscopic filaments that sprout from battery anodes during charging, are not soft and pliable at all. They are hard, rigid, and brittle. Under stress, they snap.
"Lithium dendrites have long been assumed to be soft and ductile, like Play-Doh," said Xing Liu, an assistant professor of mechanical and industrial engineering at New Jersey Institute of Technology and co-lead author of the study. "But our observations suggest that they may instead be strong and brittle, snapping more like dry spaghetti."
That distinction is not a curiosity. It changes how researchers think about why batteries fail, and what might be done to stop it.
The Invisible Problem Inside Every Battery
Dendrites have been a known hazard in lithium-based batteries for decades. They form at the anode, the negative terminal, during the charging process, growing outward in branching structures that can measure around 100 times narrower than a human hair. If a dendrite extends far enough, it can pierce the thin separator between the anode and the positively charged cathode, creating an internal short circuit. That is the scenario behind some of the most dangerous battery failures on record.
Even short of a short circuit, dendrites cause trouble. When they break off under stress, the disconnected fragments become electrically isolated from the rest of the electrode. These orphaned pieces are called dead lithium. They no longer contribute to the battery's capacity, and they accumulate over time, quietly draining the battery's ability to hold a charge.
"At present, there is no practical method to clear dendrites from a working battery cell," Liu noted.
What made dendrites so difficult to study was not just their size. Lithium reacts aggressively with air and moisture, changing its chemical and physical properties almost immediately upon exposure. Getting accurate measurements of how these structures actually behave inside a working battery required building something that had never been built before.
Building a Window Into the Nanoscale
The research brought together teams from New Jersey Institute of Technology, Rice University, Georgia Institute of Technology, the University of Houston, and Nanyang Technological University in Singapore. Their challenge was to harvest individual dendrites from real batteries and test their mechanical properties without ever letting the samples touch the air.
Researchers grew dendrites under realistic battery conditions, using copper grids as current collectors inside sealed coin cells. Once grown, the dendrites were transferred using a nanomanipulator inside a scanning electron microscope, a process so delicate that even the microscope's electron beam had to be managed carefully. The team discovered that the beam itself could alter the material's properties, softening the dendrites through a heating effect. They had to conduct the critical tensile tests with the beam switched off, tracking deformation instead through tiny marker patterns placed near each sample.
"To enable the quantitative study of lithium dendrites, we developed customized sample preparation and mechanical characterization platforms for such delicate work," said Boyu Zhang, a Rice doctoral alumnus and co-lead author on the study.
Co-lead author Qing Ai, a former research scientist at Rice University, put the significance plainly: "Despite decades of study, the fundamental nanomechanical properties of lithium dendrites remained a mystery, until now."
What the tests showed was stark. Bulk lithium yields under just 0.6 megapascals of stress and stretches up to 35 percent before breaking. Individual dendrites fractured at stresses exceeding 150 megapascals, more than 250 times stronger, with no visible stretching or deformation beforehand. The break was clean and perpendicular, the fracture surface showing no sign of the necking that ductile materials display before failure.
The Shell That Changes Everything
To understand why, the team turned to cryo-transmission electron microscopy, an imaging technique that preserves delicate samples at extremely low temperatures. What they found inside each dendrite was a core-shell structure. The lithium crystal at the center was encased in a thin outer layer called the solid electrolyte interphase, or SEI, roughly 15 nanometers thick. That coating forms naturally as the electrolyte reacts with the lithium surface during battery operation.
The SEI shell is what makes dendrites dangerous in a way nobody had fully accounted for. In a bare lithium structure at the nanoscale, plastic deformation begins when dislocations, small defects in the crystal structure, move and multiply. The surface of the material provides the easiest starting point for that process. But when the SEI encases the lithium core, it eliminates that easy starting point. The dislocation has nowhere to begin. The material becomes locked, unable to yield gradually, and instead stores stress until it shatters.
Computational modeling at NJIT and Georgia Tech confirmed this mechanism. Simulations estimated that the yield stress of the lithium core inside a dendrite exceeds 115 megapascals, a figure that would have seemed implausible for a metal as soft as lithium before this work.
"We conducted scale-bridging simulations to explain why lithium dendrites behave differently from previously thought," Liu said.
The team also tested dendrites grown in solid-state battery systems, not just those with liquid electrolytes, and observed the same brittle behavior. Real-time footage captured inside operating solid-state batteries showed dendrites fracturing under the mechanical stress of plating and stripping cycles, directly linking their brittleness to the dead lithium fragments that accumulate during normal use.
A New Map for an Old Problem
The finding reframes several puzzles that had lingered in battery research. One of the most stubborn was how soft lithium dendrites could crack solid electrolytes far harder than themselves. A brittle dendrite under pressure behaves more like a wedge than a soft probe, concentrating stress at a point and driving fractures into rigid materials. That mechanical picture makes what was previously unexplained easier to understand.
It also points toward specific strategies for addressing the problem. Because the brittleness of dendrites stems from the suppression of dislocation activity in the lithium core, one approach would be to replace pure lithium anodes with lithium alloys. Alloying introduces new dislocation sources into the material, giving the crystal somewhere to begin yielding before catastrophic fracture. That allows dendrites to deform rather than snap, reducing the buildup of dead lithium fragments and potentially lowering the risk of separator penetration.
Liu described these mechanical mechanisms as new "notes" in the larger composition of materials design for high-energy batteries.
"The strengthening mechanism we identified in lithium dendrites adds a new note to this composition," he said.
Practical Implications of the Research
The findings arrive at a moment when lithium-metal batteries are attracting serious commercial and research attention. Unlike the graphite anodes in most consumer electronics today, lithium-metal anodes can store far more energy per gram, making them attractive for electric vehicles and grid storage. Dendrite formation has been one of the primary obstacles to making those batteries viable and safe at scale.
Understanding that dendrites are brittle rather than ductile changes the engineering conversation in concrete ways. Anode materials, electrolyte formulations, and separator designs can all be reconsidered with this mechanical reality in mind.
Solid electrolyte microstructures may be tailored to resist the wedge-like cracking behavior that brittle dendrites produce. And the formation of dead lithium, previously attributed mainly to uneven electrochemical dissolution, now has a parallel mechanical explanation: physical fracture during normal charging and discharging cycles.
For researchers and engineers working to extend battery life and prevent failure, the spaghetti analogy may prove more useful than it sounds.
Research findings are available online in the journal Science.
The original story "Hard truth: Here's the reason most lithium-ion batteries fail" is published in The Brighter Side of News.
Related Stories
- Safer, low-cost alternative to lithium batteries could bring electricity to a billion underserved people
- Proton batteries offer safer, faster charging than lithium-ion
- Vegetable oil is the secret weapon in recycling lithium-ion batteries
Like these kind of feel good stories? Get The Brighter Side of News' newsletter.
Shy Cohen
Writer



